Results of an international study conducted in patients with knee OA
 A Franco-British work has been recently published in the Journal CARTILAGE(1) and presented at the 4th edition of the International Symposium on Intra-articular Treatment (ISIAT), which took place last 5th to 7th of October 2017.
A Franco-British work has been recently published in the Journal CARTILAGE(1) and presented at the 4th edition of the International Symposium on Intra-articular Treatment (ISIAT), which took place last 5th to 7th of October 2017.
This work analysed the percentage of “responders”, 6 months after a single intra-articular injection of HAppyCross, according to several Patients-Reported-Outcomes (PROs). Each patient recruited had a symptomatic knee OA and was not sufficiently improved by the first-line treatments including analgesics or non-steroidal anti-inflammatory drugs (NSAIDs).
In this study, the authors assumed that the rate of response to viscosupplementation could vary depending on the choice of the PROs and that a state-attainment criterion, such as the PASS, has to be preferred to a responder criterion, such as pain decrease, to assess the individual clinical status of a patient at a given time (here, 6 months after the injection), as it is done in daily practice. In other words, clinical response should rather be “I’m feeling good” rather than “I’m feeling better”.
An observational study with a standardized follow-up
This study was a prospective, observational study conducted in daily clinical practice. Every patient recruited was followed using a standardized procedure at baseline and month 6. The PROs were WOMAC pain and function, patient’s self-assessment of satisfaction and patient’s global assessment of pain (PGAP).
The number and percentage of patients who reached the PASS threshold (Patient Acceptable Symptom State) (PASS+) have been obtained and compared to several validated PROs previously published such as « PASS Pain», “PASS Function », and “PASS PGAP”(2) which all assess the clinical status of a patient at a given time, by classifying him or her as being in “an acceptable state”. PASS score can be subject to some variations related to the country or method of collection but all are from 3 to 4 on a 10 point rating numeric scale.
An exceptional response rate at 6 months
53 patients with symptomatic knee OA have been analysed. Chart 1 recaps the percentage of responders depending on the choice of the PROs.
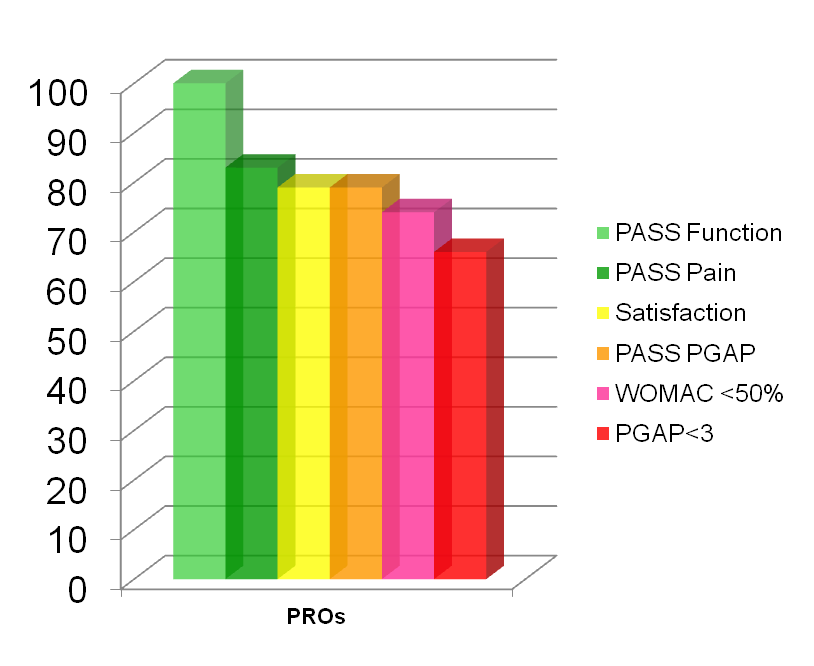
Chart 1
6 months following a single intra-articular injection of HAppyCross in the knee, 100% of the patients were” PASS + function” and 83% “PASS + pain”. “PASS + PGAP” was the one which correlated the best with patients’ level of satisfaction (79% for both outcomes) while 73.6% of patients experienced a > 50% decrease in WOMAC pain, showing that decrease of pain of more than 50% is not the only satisfaction and efficacy outcome for the patients.
Finally, authors showed that PGAP score was significantly higher than WOMAC pain and WOMAC function sub-scores as well as than the item 1, 3, 4, and 5 of WOMAC pain. However, there was no statistically significant difference between PGAP and item 2 of WOMAC pain (pain at stair climb up and down), suggesting that pain score reported by patients in PGAP is mainly due to pain occurring in the most painful situation such as going up and down the stairs. Consequently, all patients who had a PGAP ≤3/10 at 6 month were satisfied with the treatment.
It should be noted that, as previously demonstrated, a higher BMI was associated with a lower rate of PASS+ (P = 0.1) and the difference was more significant between obese (BMI>30 kg/m2) and non-obese subjects (P = 0.03).
Tolerance of the treatment has been excellent, confirming results of previous studies (3, 4, 5). Only 3 patients experienced transient worsening of pain in the target knee which occurred a few hours after injection and resolved without sequel between 36 and 72 hours.
This prospective study shows that 8 patients out of 10 are satisfied 6 months after a single intra-articular injection of HAppyCross in the knee.
HAppyCross innovative and unique formulation, which combines highly concentrated and cross-linked HA with high concentration of mannitol (77mg) in only 2.2ml, explains the excellent benefit/risk ratio of a single intra-articular injection of HAppyCross in patients suffering from knee OA.
.
![]()
Read the Abstract
.
.
.
![]()
Read Full Text
References
1- Conrozier T, Monet M, Lohse A, Raman R. Getting Better or Getting Well? The Patient Acceptable Symptom State (PASS) Better Predicts Patient’s Satisfaction than the Decrease of Pain, in Knee Osteoarthritis Subjects Treated with Viscosupplementation. Cartilage. 2017 Aug 1:1947603517723072.
2- Bellamy N et al. Development of multinational definitions of minimal clinically important improvement and patient acceptable symptomatic state in osteoarthritis. Arthritis Care Res [Hoboken]. 2015;67[7]:972-80.
3- Conrozier T et al. Standardized Follow-up of Patients with Symptomatic Knee Osteoarthritis Treated with a Single Intra-articular Injection of a Combination of Cross-Linked Hyaluronic Acid and Mannitol. Clin Med Insights Arthritis Musculoskelet Disord. 2016; 25;9:175-9. doi: 10.4137/CMAMD.S39432.
4- Henrotin Y et al. Reduction of the Serum Levels of a Specific Biomarker of Cartilage Degradation (Coll2-1) by Hyaluronic Acid (KARTILAGE® CROSS) Compared to Placebo in Painful Knee Osteoarthritis Patients: the EPIKART Study, a Pilot Prospective Comparative Randomized Double Blind Trial. BMC Musculoskelet Disord. 2017;18(1):222
5- Monet M et al. Safety and Efficacy of Single Intra-Articular Injection of a Cross-Linked Hyaluronic Acid/Mannitol Formulation [Happycross®] in knee Osteoarthritis Results of a Prospective Observational Study in Daily Practice Conditions. Ortho & Rheum Open Access 2017; 5(3): 555664



